Water Composition (1/5)

We seldom consider the water we dive in, yet it is a fascinating subject in its own right.
Basic Chemistry
Water is an odorless, tasteless, transparent liquid that is colorless in small amounts but exhibits a bluish tinge in large quantities. It is the most familiar and abundant liquid on earth. In solid form ( ice ) and liquid form it covers about 70% of the earth's surface. It is present in varying amounts in the atmosphere. Most of the living tissue of a human being is made up of water; it constitutes about 92% of blood plasma, about 80% of muscle tissue, about 60% of red blood cells, and over half of most other tissues. It is also an important component of the tissues of most other living things.
The word water comes from the Old English waeter; German Wasser, originally from hypothetical Proto-Indo-European wod-or.
compiled from various sources
Chemical and Physical Properties
Chemically, water is a compound of hydrogen and oxygen, having the formula H2O. It is chemically active, reacting with certain metals and metal oxides to form bases, and with certain oxides of nonmetals to form acids. It reacts with certain organic compounds to form a variety of products, e.g., alcohols from alkenes. Because water is a polar compound, it is a good solvent. Although completely pure water is a poor conductor of electricity, it is a much better conductor than most other pure liquids because of its self-ionization, i.e., the ability of two water molecules to react to form a hydroxide ion, OH-, and a hydronium ion, H3O+. Its polarity and ionization are both due to the high dielectric constant of water.
Water has interesting thermal properties. When heated from 0°C, its melting point, to 4°C, it contracts and becomes more dense; most other substances expand and become less dense when heated. Conversely, when water is cooled in this temperature range, it expands. It expands greatly as it freezes; as a consequence, ice is less dense than water and floats on it. Because of hydrogen bonding between water molecules, the latent heats of fusion and of evaporation and the heat capacity of water are all unusually high. For these reasons, water serves both as a heat-transfer medium ( e.g., ice for cooling and steam for heating ) and as a temperature regulator ( the water in lakes and oceans helps regulate the climate. )
Structure of the Water Molecule
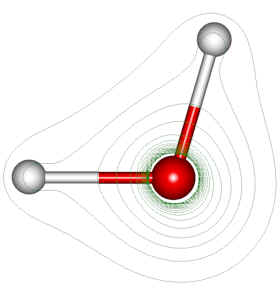
Many of the physical and chemical properties of water are due to its structure. The atoms in the water molecule are arranged with the two H-O bonds at an angle of about 105 degrees rather than on directly opposite sides of the oxygen atom. The asymmetrical shape of the molecule arises from a tendency of the four electron pairs in the valence shell of oxygen to arrange themselves symmetrically at the vertices of a tetrahedron around the oxygen nucleus. The two pairs associated with covalent bonds holding the hydrogen atoms are drawn together slightly, resulting in the angle of 105 degrees between these bonds. This arrangement results in a polar molecule, since there is a net negative charge toward the oxygen end ( the apex ) of the V-shaped molecule and a net positive charge at the hydrogen end. The electric dipole gives rise to attractions between neighboring opposite ends of water molecules, with each oxygen being able to attract two nearby hydrogen atoms of two other water molecules. Such hydrogen bonding, as it is called, has also been observed in other hydrogen compounds. Although considerably weaker than the covalent bonds holding the water molecule together, hydrogen bonding is strong enough to keep water liquid at ordinary temperatures; its low molecular weight would normally tend to make it a gas at such temperatures otherwise.
Various other properties of water, such as its high specific heat, are due to these hydrogen bonds. As the temperature of water is lowered, clusters of molecules form through hydrogen bonding, with each molecule being linked to others by up to four hydrogen bonds, each oxygen atom tending to surround itself with four hydrogen atoms in a tetrahedral arrangement. Hexagonal rings of oxygen atoms are formed in this way, with alternate atoms in either a higher or lower plane than their neighbors to create a kinked three-dimensional structure.
Liquid Water
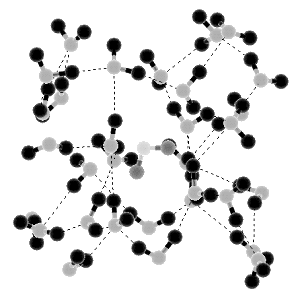
According to present theories, water in the liquid form contains three different molecule populations. At the highest temperatures single molecules are the rule, with little hydrogen bonding because of the high thermal energy of the molecules. In the middle range of temperatures there is more hydrogen bonding, and clusters of molecules are formed. At lower temperatures aggregates of clusters also form, these aggregates being the most common arrangement below about 15°C.
On the basis of these three population types and the transitions between them, many aspects of the anomalous behavior of water can be explained. For example, the tendency of water to freeze faster if it has been cooled rapidly from a relatively warm temperature than if it has been cooled at the same rate from a lower temperature is explained in terms of the greater number of irregularly shaped cluster aggregates in the cooler water that must find a suitable means of fitting together with a neighboring aggregate.
Ice
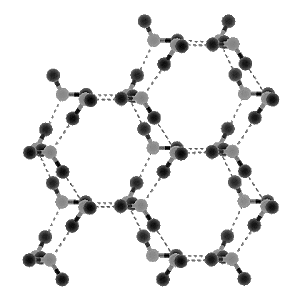
In ice, each molecule forms the maximum number of hydrogen bonds, resulting in crystals composed of open, hexagonal columns. Because these crystals have a number of open regions and pockets, normal ice is less dense than water. However, other forms of ice also exist at conditions of higher pressure, each of these different forms ( designated ice II, ice III, etc. ) having greater density and other distinct physical properties that differ from those of normal ice, or ice I. As many as eight different forms of ice have been distinguished in this manner. * the higher pressures creating such forms cause rearrangements of the hexagonal columns in ice, although the basic kinked hexagonal ring is common to all forms.
When ice melts, it is thought that the fragments of these structures fill many of the gaps that existed in the crystal lattice, making water denser than ice. This tendency is the dominant one between 0°C and 4°C, at which temperature water reaches its maximum density. Above this temperature, expansion due to the increased thermal energy of the molecules is the dominant factor, with a consequent decrease in density.
* Editor's note: I recall once reading a sci-fi short story in which scientists created "Ice IX" - the ninth form of ice. This artificial ice had two interesting properties: first, it was its own catalyst - self-reproducing, and second, it could form at temperatures well above freezing. Of course, when it got out of the lab, the whole earth "froze".
Density
Pure liquid water has a density of 62.4 pounds per cubic foot ( or 8.341pounds per gallon ) at a reference temperature of 39°F. Over a biologically relevant range of temperatures, the density of pure liquid water ranges from 62.42 lbs/cu.ft at 32°F to about 62.0 lbs/cu.ft at 85°F. The density of seawater is somewhat higher owing to the dissolved salts, ranging from 64.1 to 64.9, with a standard value of 64.6 at 39°F. The density of seawater is highly dependent on the salinity.
The density of ice is considerably less at 56.2 pounds per cubic foot; water being one of the few liquids that shows a decrease in density upon solidifying. Thus, ice floats. While this was unfortunate for the Titanic, it is otherwise a very lucky thing for life on earth. If ice did not float, all bodies of water would freeze solid from the bottom up, and life as we know it would have a very difficult time surviving! As it is, a winter surface layer of ice and snow actually insulates the liquid water below and protects the life in it.
Viscosity
| Temperature ( oF ) | Absolute Dynamic Viscosity ( cp ) |
| 32 | 1.794 |
| 40 | 1.546 |
| 50 | 1.310 |
| 60 | 1.129 |
| 70 | 0.982 |
| 80 | 0.862 |
| 90 | 0.764 |
Viscosity is the measure of a liquid's resistance to flow, or its opposition to the movement of an object through it. High viscosity liquids are "thick", like syrups and motor oil. Water is a fairly low viscosity liquid. The viscosity of water varies with temperature and density, although the temperature variation is far greater than the density variation.
The viscosity of freshwater over a range of biologically relevant temperatures is shown at right. Saltwater is slightly higher. Without going into too much detail, it is apparent that cold water is approximately twice as viscous as warm water, or that drag in cold water is much greater than in warm water.
This in turn explains why warm water fishes can have such outlandish shapes, while cold water fishes are all much more streamlined and boring. This may also explain why you can swim faster in the Bahamas than in New Jersey, although I think a drysuit and double tanks might also have something to do with it.
Specific Heat
Specific heat describes the relationship of temperature to heat for a given substance. Water, with a specific heat of 4.184 J/g-deg C has one of the highest specific heats of any substance. This property of water allows it to absorb or release large amounts of heat without changing its temperature dramatically. This is a very important feature, since it allows the large bodies of water on the earth's surface to moderate our climate so that temperatures do not get too hot or too cold. Water also has a very high heat of vaporization, and freezing. All of these factors combine to make the aquatic environment much more temperature-stable than our own terrestrial conditions. Marine organisms especially are adapted to fairly steady temperature conditions, and are often killed by abrupt changes in temperature that we would take in stride.

